Forth Valley patients in 'life saver' drug research to help seriously ill COVID cases
and live on Freeview channel 276
And the people who took part in the Recovery Trial - some of them seriously ill - have been thanked for their “courage and willingness.”
A total of 77 people were recruited by doctors visiting wards in Forth Valley Royal Hospital who discussed the initiative with their patients.
Advertisement
Hide AdAdvertisement
Hide AdThe Recovery trial has been testing a range of potential treatments for Covid-19 since last March when the pandemic first impacted.
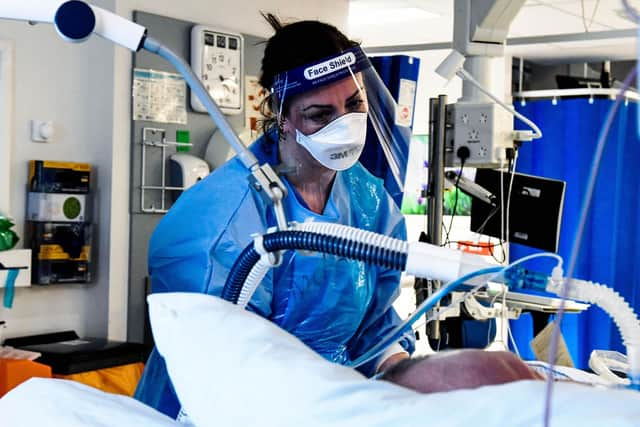

Tocilizumab, an intravenous drug used to treat rheumatoid arthritis, was added to the trial in April 2020 for patients with COVID-19 who required oxygen and had evidence of inflammation.
The hospital continued to recruit until the end of January 2021 when, in the view of the trial Steering Committee, sufficient patients had been enrolled to establish whether or not the drug had a meaningful benefit.
Now, the first results have been revealed - and they are positive.
Advertisement
Hide AdAdvertisement
Hide AdThe results showed that Tocilizumab reduces mortality by about one third for patients requiring simple oxygen - and nearly one half for those requiring invasive mechanical ventilation.
It also shortens the length of time patients stay in hospital until they are able to be discharged.
The findings were warmly welcomed by clinicians.
Dr Mark Spears, NHS Forth Valley consultant in respiratory medicine, said: “This is an excellent result for those looking for additional treatments for Covid-19.
“Tocilizumab, in addition to dexamethasone, will now be provided early to suitable patients on presentation to hospital, reducing their need for intensive care, improving their chances of survival and reducing time to discharge.”He paid tribute to everyone who took part.
Advertisement
Hide AdAdvertisement
Hide AdDr Spears added: “I would like to thank all those involved in the trial to date, including the trials pharmacy team, the research nurses, clinicians looking after patients with Covid-19 and most importantly the patients themselves.
“Without their courage and willingness to take part in clinical trials, even when seriously unwell, we would be unable to find new effective treatments for this horrible disease.”